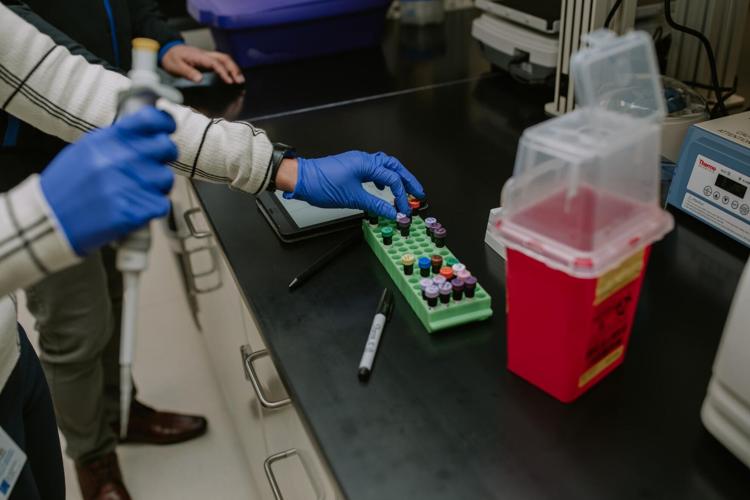
For Dr. Shernan Holtan, Roswell Park Comprehensive Cancer Center’s newest cell therapy and transplant expert, there is a unique sense of urgency when it comes to her team’s work.
Why? She’s already seen it drastically alter the course of transplant treatment as we know it.
“The last time our field changed this substantially was 1986,” she says. “It’s not like people hadn’t tried. A lot of studies had come and gone – just nothing that moved the needle like this did.”
Defeating transplant complications
Dr. Holtan and Dr. Brian Betts, who has worked at Roswell Park since August 2023, previously worked together at the University of Minnesota, where they researched ways to improve outcomes for stem cell transplant patients, specifically those suffering a life-threatening complication called graft versus host disease (GvHD).
“This used to be a complication that would actually lead people to not want to even undergo a transplant,” Dr. Holtan explains. “It’s a complication where the donor immune system attacks the patient.”
Their research, however, transformed the way GvHD is treated, turning it from a highly feared complication that around 60% of patients experienced into a rarity.
“Less than 5% of people now experience significant graft versus host disease, and it’s rarely fatal,” Holtan says. “Now that we’re seeing this reduced risk, we’re able to focus on other issues that can come up after transplant. And that’s the momentum that we’ll be continuing here.”
Those other issues include the risk of cancer relapse and the long-term effects, which can include high medication loads and months of recovery. Dr. Holtan is interested in leveraging the type of cutting-edge research she and Dr. Betts have pioneered over the last several years and applying it to these new fronts, making transplant a less burdensome treatment overall.
“I’m just hoping that we can expand access and make it so that more patients potentially have this life-saving therapy,” she says.
What’s next for cell therapy

Next on the CAR T horizon? Finding ways to make them more targeted and resilient, and as effective against cancers involving solid tumors as they are against leukemias, lymphomas and multiple myelomas.
This is just one area of research for Roswell Park’s cell therapy and transplant research. Cell therapy, which uses living cells to destroy and control cancer cells, also involves ground-breaking chimeric antigen receptor T-cell (CAR T) therapies that are genetically engineered to attack cancer cells.
There are FDA-approved CAR T therapies for several cancers, including leukemias, lymphomas and multiple myelomas, some of which were pioneered by experts currently on Roswell Park’s team.
The next frontier is further honing of the CAR T therapies so they’re even more targeted and resilient as well as effective against a wider range of cancers, particularly those involving solid tumors in organs like the breast, ovaries, prostate or other body sites.
Dr. Betts says the team members’ unique positions as both laboratory scientists and physicians who see patients offer them an advantage.
“It gives you this neat opportunity to bring problems from the bedside to the lab, fix it, and then bring it back to the patient,” he says. “I think that’s pretty powerful.”
Cutting-edge facilities
Roswell Park has invested in the essential workspace to accommodate all this innovative research. So-called “clean rooms,” or Good Manufacturing Practice (GMP) facilities, are highly regulated and equipped to genetically reengineer human cells fit for use in medical treatment.
The comprehensive cancer center currently has six clean rooms and is constructing an entire floor of them to triple their capacity, set to open in 2025.
“They’re basically essential for any type of cell therapy,” Betts says. “That includes processing stem cells for a transplant and includes the engineering of the CAR T cells. Having multiples of those makes it so we’re not restricted in the lab.”
Having those facilities integrated into the campus allows a higher level of efficiency and more specialized care at all levels.
That investment in space, experts and care is a big part of why Dr. Holtan, Dr. Betts and others joined the Roswell Park team.
“There’s a lot of institutional experience, but there’s also the courage to move forward,” Holtan says. “There’s an environment of innovation. We’re not shying away from new ideas or testing new practices—the ideas are welcome.”
Meet the innovation hub
Roswell has made it a mission to assemble the best minds in cell therapy research and patient care. Over the last five years, they’ve built a team of some of the leading physician-scientists in the field, all dedicated to continuing innovation in new therapies and ensuring existing ones are as safe and effective as possible.

Renier Brentjens, MD, PhD
Deputy Director and Chair of Medicine
Arrival at Roswell Park: September 2021
Total years of experience in cell therapy: 25
Special area of expertise/interest: Part of the team that developed the first approved CAR T (chimeric antigen receptor T-cell) therapy; now focused on “armored” CAR therapies and use of CAR T to treat solid-tumor cancers

Marco Davila, MD, PhD
Senior Vice President and Associate Director for Translational Research
Arrival at Roswell Park: July 2022
Total years of experience in cell therapy: 18
Special area of expertise/interest: Preclinical development and clinical investigation of novel cell therapies for cancer. Improving outcomes and quality of life for patients receiving cell therapy as standard of care

Anuradha Krishnamurthy, MBBS
Associate Director of Early Phase Clinical Trials for Solid Tumor Cell Therapies
Arrival at Roswell Park: September 2022
Total years of experience in cell therapy: 10
Special areas of expertise/interest: Expanding the benefits of T-cell therapies to patients with solid-tumor cancers such as colon, pancreatic, breast, ovarian, etc.

Peter Maslak, MD
Vice Chair of Clinical Affairs and Technical Director, Clinical Flow Cytometry Laboratory
Arrival at Roswell Park: March 2023
Total years of experience in cell therapy: 33
Special area of expertise/interest: Monitoring immune response and use of vaccination and cell-based immunotherapies supporting new, more effective cancer treatments; new treatments for leukemia

Yeong “Christopher” Choi, PhD, MBA
Senior Vice President of Industry Partnerships and Technical Director, Roswell Park GMP Engineering & Cell Manufacturing Facility
Arrival at Roswell Park: 2010-2018; rejoined March 2023
Total years of experience in cell therapy: 21
Special area of expertise/interest: Cell and gene therapy, Good Manufacturing Practice (GMP) manufacturing, business operations, quality and process development

Kanwaldeep Mallhi, MD
Clinical Director of Pediatric Transplantation and Cellular Therapy, Department of Pediatrics
Arrival at Roswell Park: August 2023
Total years of experience in cell therapy: 10
Special area of expertise/interest: New approaches using stem cell transplant, gene therapy and cell therapy to improve curative outcomes for children with cancer and nonmalignant disorders

Brian Betts, MD, PhD
Vice Chair of Strategic Initiatives in Transplant & Cellular Therapy
Arrival at Roswell Park: August 2023
Total years of experience in cell therapy: 16
Special area of expertise/interest: Developing new engineered T-cell therapies for cancer; ways to prevent or reduce graft versus host disease (GvHD) and other complications of stem cell transplant

Shernan Holtan, MD
Chief of Blood and Marrow Transplantation
Arrival at Roswell Park: February 2024
Total years of experience in cell therapy: 15
Special area of expertise/interest: Reducing toxicities of transplantation, prevention/treatment of graft-versus-host disease, nonmalignant and immune-based diseases, prevention of sarcopenia/strength training